The laboratory of intestinal adaptation and regeneration
R&D > Laboratories > The laboratory of intestinal adaptation and regeneration – Prof. Igor Sokhotnik >>

Our Vision
Our vision is to evaluate signaling pathways involved in intestinal stem cell fate decision following intestinal ischemia-reperfusion in a rat and in Caco-2 cells. Also we are planning to Investigate the role of signaling pathways in regulation of stem cell activity in neonates with NEC compared to control group without a disease.
We will continue to evaluate pathways related to bowel motility after massive resection of the small bowel in a rat and we are planning to use stem cells to encourage the growth and recovery of intestinal cells after massive small bowel resection in a rat.

Contact Us
Primary Investigators

Prof. Igor Sokhotnik, Lab PI
Phone: +972-3-697-----
Email: igorsu@tlvmc.gov.il

Yulia Pollak, Lab manager
Phone: +972-3-697----
Email: yuliapo@tlvmc.gov.il
Address
Sammy Ofer Heart Building
10th floor 6 weizmann st., Tel Aviv

Research
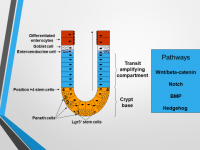
Intestinal stem cells (ISCs) are fundamental cornerstones in intestinal biology, ensuring homeostatic self-renewal of intestinal mucosa and presenting a reserve pool of cells that can be activated after tissue injury or after bowel resection. Intestinal stem cells are characterized by their ability to self-renew as well as to differentiate into specialized cells, properties critical for tissue maintenance and regeneration.
The regulation of ISC is made by four main signaling pathways Wnt/β-catenin, Hedgehog, Bone morphogenetic protein (BMP) and Notch. We are investigating the roles of these signaling cascades in stimulation of cell proliferation and differentiation following intestinal IR and massive small bowel resection in a rat model which are unknown.
Extracellular-vesicles (EVs) are membrane vesicles secreted by various types of cells and controlling multiple cell processes. Placental human villous trophoblasts (HVT)-EV are sheds from trophoblasts cells during pregnancy, contained growth factors and miRNA.
Necrotizing enterocolitis (NEC) and neonatal chronic lung disease (CLD) are devastating diseases that affects preterm neonates, characterized by high morbidity and mortality.
We are evaluating the effects of HVT-EVs treatment effects on in-vivo models: a). NEC rat model. b). CLD rat model (in collaboration with the Hematology Research Laboratory Tel Aviv Sourasky Medical Center)
Gallery



Our Team
Current Staff
Researchers
- Dr. Yoav Ben-Shahar
- Dr. Odelia Eshel-Furer
- Dr. Keren Kremer
Past Staff
Current funding


Highlighted Publications
Anti-TNF-α Therapy Exerts Intestinal Anti-inflammatory and Anti-Apoptotic Effects After Massive Bowel Resection in Rats. Sukhotnik I, Ben-Shahar Y, Pollak Y, Cohen S, Guz-Mark A, Shamir R. |
J Pediatr Gastroenterol Nutr. 2021 Jan 1;72(1):49-55. doi:10.1097/MPG.0000000000002876.PMID: 32740515.
Sukhotnik I, Ben-Shahar Y, Pollak Y, Cohen S, Moran-Lev H, Koppelmann T, Gorenberg M. Am J Physiol Gastrointest Liver Physiol. 2020 Dec 16. doi: 10.1152/ajpgi.00223.2020. Online ahead of print.PMID: 33325807 |
Koppelmann T, Pollak Y, Ben-Shahar Y, Gorelik G, Sukhotnik I. |
Nutrients. 2021 Mar 10;13(3):888. doi: 10.3390/nu13030888.PMID: 33801889