The Genomic Research Laboratory for Lung Fibrosis
R&D > Laboratories > The Genomic Research Laboratory for Lung Fibrosis- Dr. Avraham (Rami) Unterman

Our Vision
To uncover the basic mechanisms underlying lung fibrosis and develop new treatments -
Interstitital lung diseases (ILDs) consist of over 200 different diseases. Many patients, in particular those who are diagnosed with idiopathic pulmonary fibrosis (IPF), develop severe lung scarring (called lung fibrosis) that necessitates lung transplantation for survival, including some who recover from COVID-19. Despite multidisciplinary approaches to diagnose ILD patients, in many cases the exact diagnosis is unclear. Furthermore, current treatment options may delay disease progression but cannot provide cure. In our laboratory, we employ cutting-edge genomic technologies, including single cell RNA sequencing (scRNA-seq), to study ILD patient-derived samples. We believe that by using scRNA-seq, we may come one step closer towards elucidating the biology of these intricate diseases, identify new biomarkers and develop novel treatments for ILD patients.

Contact Us
Primary Investigators

Dr. Avraham (Rami) Unterman, MD, MBA, Lab PI
Head of the Multidisciplinary Pulmonary Fibrosis Service and PI of the Genomic Research Laboratory for Lung Fibrosis,Pulmonary Institute 03-6972552 ramiu@tlvmc.gov.il

Yan Stein, PhD, Lab manager
03-6972552 yanst@tlvmc.gov.il
General Contact
Address
Sammy Ofer Heart Building
10th floor Room 64-65

Research
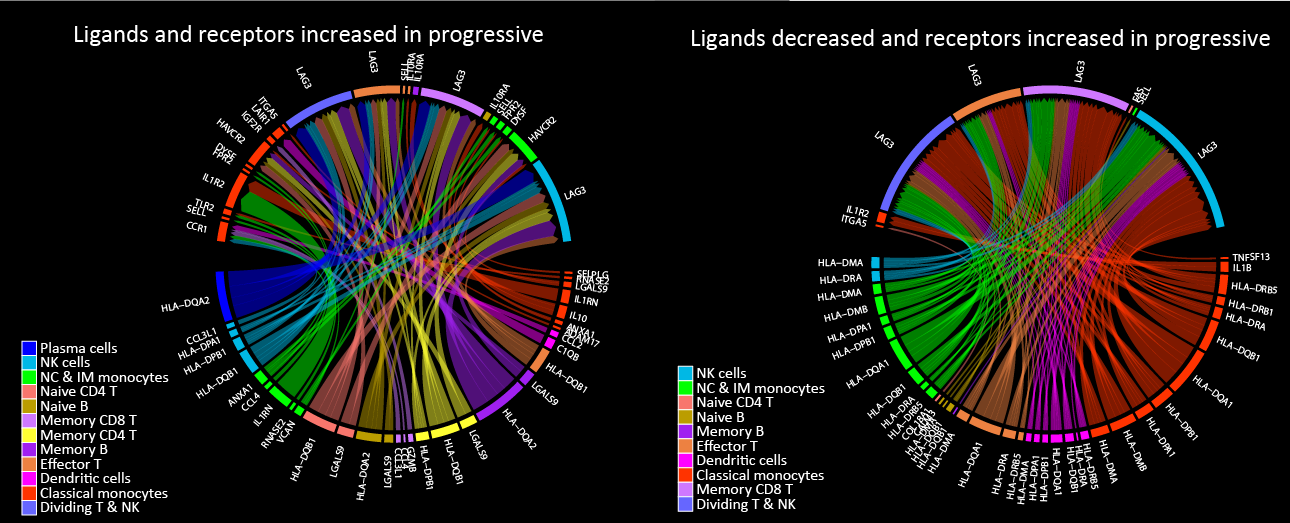
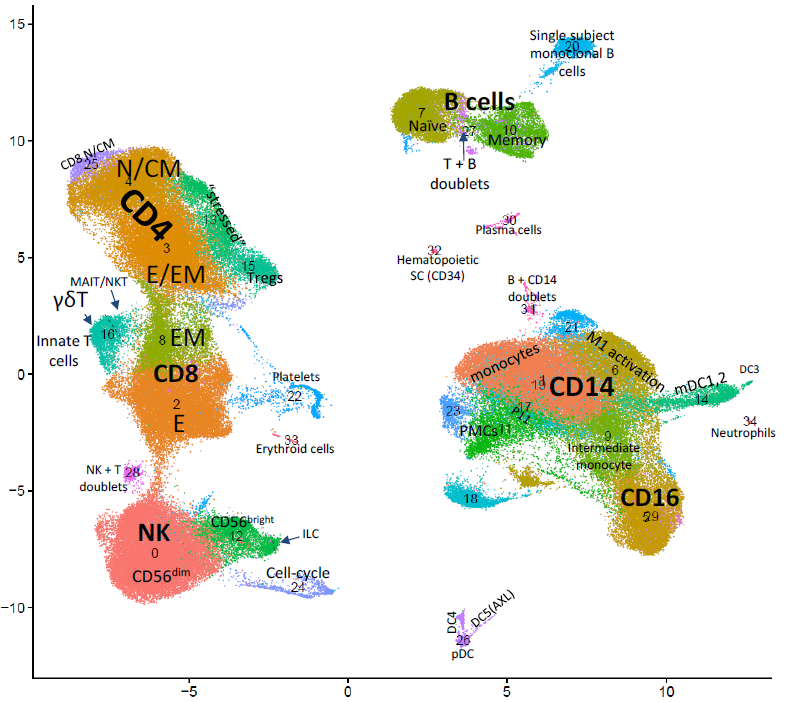
COVID-19 may manifest itself as a mild to moderate disease, or as a severe disease requiring intensive care unit (ICU)-level care that may include mechanical ventilation and extracorporeal membrane oxygenation (ECMO). Evidence suggest that dysregulated inflammation plays an important role in the exacerbation of the disease, which may lead to an increased morbidity and mortality of the disease. Nonetheless, the exact molecular mechanisms accounting for this immune dysregulation, as well as their modulation during different stages of the disease, are not fully characterized. In a joined effort together with five different laboratories in Yale School of Medicine, we performed an in-depth, single-cell multi-omics analysis of immune cells that were derived from progressive and stable COVID-19 patients. This analysis revealed unique cell populations of both the innate and adaptive immune cells, as well as aberrant cell-cell interactions between them, that characterized progressive COVID-19 patients. In addition, unique profiling of T cells and B cells revealed a skewed expansion of specific clones that characterized progressive patients. To conclude, this study revealed a dyssynchrony of the innate and adaptive immune system in progressive COVID-19 patients.
Among of the most common ILDs are idiopathic lung fibrosis (IPF), chronic hypersensitivity pneumonitis (CHP) and connective tissue-related ILDs (CTD-ILDs). It is important to differentiate between these diagnoses, since CHP and CTD-ILD require immunosuppressive therapies, while IPF is exacerbated by this treatment regime and require anti-fibrotic agents instead. We wish to analyze immune cells, which can be easily extracted from patients’ blood, by utilizing scRNA-seq. This analysis will facilitate the identification of immunological mechanisms that drive these diseases and allow the development of precision medicine approaches for diagnosis and treatment
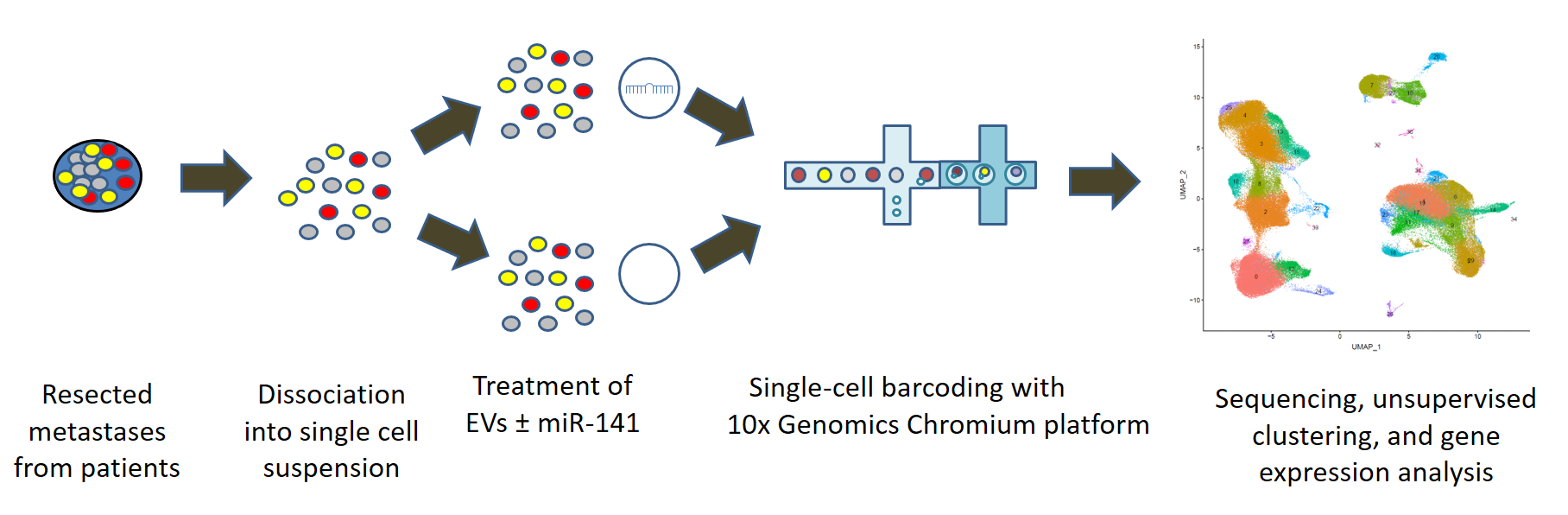
Pancreatic cancer is among the leading causes of cancer-related mortality, in which omental metastasis is a frequent event that is associated with very poor prognosis. In this study, we will examine the effects of EVs enriched with miR-141 on pancreatic cancer patient-resected metastases, as a potentially novel therapeutic approach. We will leverage on single-cell RNA sequencing in order to examine the intricate tumor-stroma interactions and biological mechanisms that govern the propagation of these metastases, as well as the potential therapeutic effect of EVs. This project is performed in collaboration with Prof. Guy Lahat and Dr. Shelly Loewenstein from the surgical oncology research laboratory.
Gallery








Our Team
Current Staff
Researchers
- Dr. Avraham (Rami) Unterman, PI
- Yan Stein, PhD, Lab Manager
- Tali Bar-Lev, PhD
Clinical Research Coordinator
- Liya Achdoot
Research Assistant
- Tzlil Hershko
Current funding




Highlight Publications
Unterman A, Sumida TS, Nouri N, Yan X, Zhao AY, Gasque V, Schupp JC, Asashima H, Liu Y, Cosme C Jr, Deng W, Chen M, Raredon MSB, Hoehn KB, Wang G, Wang Z, DeIuliis G, Ravindra NG, Li N, Castaldi C, Wong P, Fournier J, Bermejo S, Sharma L, Casanovas-Massana A, Vogels CBF, Wyllie AL, Grubaugh ND, Melillo A, Meng H, Stein Y, Minasyan M, Mohanty S, Ruff WE, Cohen I, Raddassi K; Yale IMPACT Research Team, Niklason LE, Ko AI, Montgomery RR, Farhadian SF, Iwasaki A, Shaw AC, van Dijk D, Zhao H, Kleinstein SH, Hafler DA, Kaminski N, Dela Cruz CS. Nat Commun. 2022 Jan 21;13(1):440.
Blood Transcriptomic Predicts Progression of Pulmonary Fibrosis and Associates Natural Killer Cells. SHuang Y, Oldham JM, Ma SF, Unterman A, Liao SY, Barros AJ, Bonham CA, Kim JS, Vij R, Adegunsoye A, Strek ME, Molyneaux PL, Maher TM, Herazo-Maya JD, Kaminski N, Moore BB, Martinez FJ, Noth I.
Am J Respir Crit Care Med 2021 (In Press).
Immune dysregulation and autoreactivity correlate with disease severity in SARS-CoV-2-associated multisystem inflammatory syndrome in children. Ramaswamy A, Brodsky NN, Sumida TS, Comi M, Asashima H, Hoehn KB, Li N, Liu Y, Shah A, Ravindra NG, Bishai J, Khan A, Lau W, Sellers B, Bansal N, Guerrerio P, Unterman A, Habet V, Rice AJ, Catanzaro J, Chandnani H, Lopez M, Kaminski N, Dela Cruz CS, Tsang JS, Wang Z, Yan X, Kleinstein SH, van Dijk D, Pierce RW, Hafler DA, Lucas CL
Immunity 2021; 54(5):1083-1095.e7.
More Publications >>
. Cutting Edge: Distinct B Cell Repertoires Characterize Patients with Mild and Severe COVID-19 Hoehn KB, Ramanathan P, Unterman A, Sumida TS, Asashima H, Hafler DA, Kaminski N, Dela Cruz CS, Sealfon SC, Bukreyev A, Kleinstein SH
J Immunol 2021;206(12):2785-2790.
. Type I Interferon Transcriptional Network Regulates Expression of Coinhibitory Receptors in Human T cells Sumida TS, Dulberg S, Schupp J, Stillwell HA, Axisa PP, Comi M, Lincoln M, Unterman A, Kaminski N, Madi A, Kuchroo VK, Hafler DA
BioRxiv 2020 (pre-print). Under review in Nature Immunology
Less Publications >>